Chimeric antigen receptor (CAR) T cell therapy has emerged as one of the major breakthroughs in cancer immunotherapy in the last decade. Clinical trials with autologous CAR-T cells have shown very promising results and led to the approval of numerous CAR-T cell therapies. Until now, the use of autologous T cells to manufacture CAR products has become the norm, however, recent evidence has demonstrated T-cell dysfunction in some cancer patients, which impairs the effectiveness of the end CAR-T product. Nonetheless, the use of allogeneic CAR-T cells from donors has many potential advantages over autologous approaches.
Universal or allogeneic CAR-T cells from healthy donors, provide many advantages over autologous T cells and are a potentially viable source of functional cells. Allogeneic CAR-T cells have the advantage of being ready-to-use and can be readily administered to patients from the moment of diagnosis.
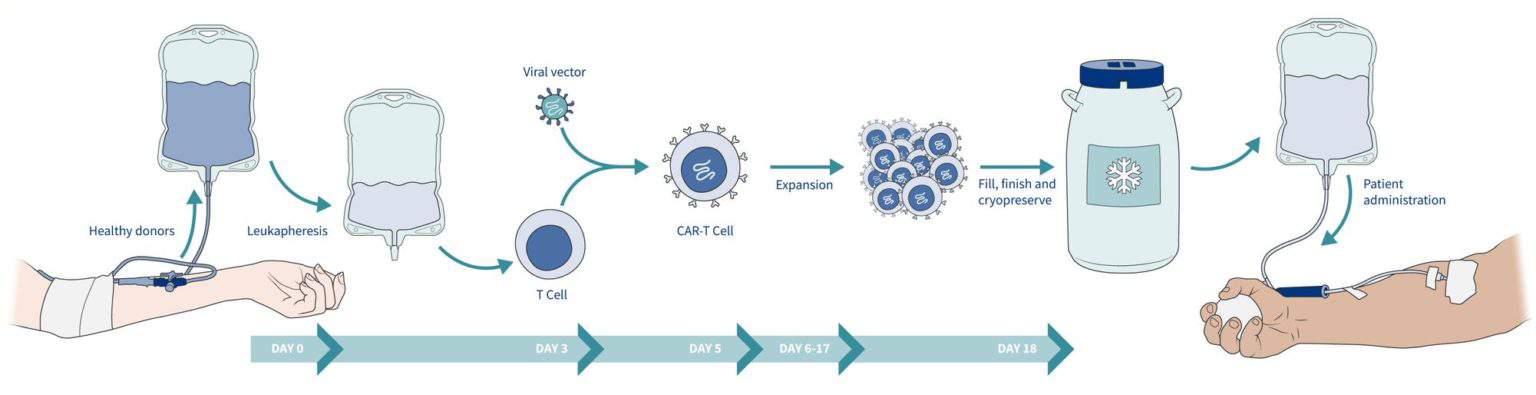
With access to a diverse pool of reliable donors from one of the largest donor blood banks in the world, Excellos donors can be targeted to expediate the development of your allogenic CAR-T.
Our data-driven donor characterization and integration system takes donor annotation to the next level. As each of our donors arrive at the donor center, samples are collected and analyzed to characterize their cells and drive manufacturing decisions. Our algorithm also correlates data from decades of donor activity across various collection types to build a heavily annotated profile for any given donor. This, combined with the Excellos’ next level functional characterization data, including FACs analysis and functional assays, helps us better predict which donors will best match your CAR-T therapy goals.
Having access to high-quality leukapheresis cellular starting material is a critical step in the success of all CAR-T cell therapies. In fact, high-quality cellular starting material has been proven to contribute to the quality of the product downstream. However, at Excellos, we understand that it’s not enough to have just have access to large and diverse donor pool, with the ability to recall donors, technical expertise of the apheresis staff and scientists with decades of experience is needed in order to process the collections effectively. Post-thaw viability of cellular material must remain high if you expect the cells to perform well during manufacturing.
High quality cellular starting material is critical in all stages of CAR-T development, from discovery through commercialization, as it contributes to the quality of the product downstream. To support your CAR-T needs, Excellos provides highly characterized leukopaks from healthy donors. Combining our scientific expertise and extensive functional characterization creates an annotation platform that can help inform manufacturing decisions, allowing you to select the best donor for the development of your allogenic CAR-T.
Contact Us to Order your Cellular Starting Material
All leukapheresis products are obtained by apheresis of normal donors in our FDA-registered donor center. All donors (collected under IRB-approved consent) undergo testing for infectious disease with common blood-borne pathogens and viruses including RPR, HIV, LCMV, and HVC. Excellos donors are also extensively characterized (functionally, phenotypically and metabolically). Apheresis collections from healthy donors are shipped fresh, cryopreserved, or purified and isolated to PBMCs under cGMP guidelines.
Excellos Quality Control – To ensure viability and cell counts of our cellular products, we implement random sampling per lot. A Certificate of Analysis (CoA) is included with every shipment. Characterization by immunophenotyping can also be performed upon request and included in the CoA.